[ad_1]
New research has uncovered five genes linked to Lewy Body dementia, which supports the association between that condition and both Parkinson’s disease and Alzheimer’s disease.
The study, led by researchers from the US National Institutes of Health, was published online February 15 in Nature Genetics.
“Lewy body dementia is a devastating brain disorder for which we have no effective treatments. Patients often appear to suffer the worst of both Alzheimer’s and Parkinson’s diseases,” said senior author Sonja Scholz, MD, PhD.
“Our results support the idea that this may be because Lewy body dementia is caused by a spectrum of problems that can be seen in both disorders,” she said. “We hope that these results will act as a blueprint for understanding the disease and developing new treatments.”
For the study, the researchers compared the chromosomal DNA sequences of 2981 patients with Lewy body dementia with those of 4391 healthy, age-matched control participants. Samples were collected from participants of European ancestry at 44 sites (17 in Europe and 27 across North America).
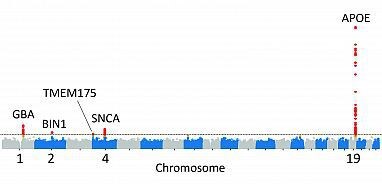
Sequences of five genes may help determine whether a person will suffer from Lewy body dementia. Credit: Scholz lab, NIH/NINDS
“We found strong genetic evidence that variants in the genes GBA, BIN1, TMEM175, SNCA-AS1, and APOE influence the risk for developing Lewy body dementia,” Scholz, chief of the neurodegenerative diseases research unit at the National Institute of Neurological Disorders and Stroke (NINDS), told Medscape Medical News.
It was the first time that two of the genes — BIN1 and TMEM175 — had been implicated in the disease. The other three genes, SNCA, APOE, and GBA, had been associated in previous studies, and thus, strengthened the importance of the genes in Lewy body dementia, the researchers report.
They also validated their results in a second analysis in which they saw differences in the same five genes in another 970 patients with Lewy body dementia vs a new set of 8928 control subjects.
Research into the exact functions of these common risk variants is still ongoing, Scholz noted. “What we know at the present time is that they are implicated in fundamental cellular processes, such as protein trafficking, protein degradation, energy and lipid metabolism, and others,” she said. “The end result is a loss of neuroprotective functions and gain of toxic functions that culminate in protein build-up, failure of mitochondrial respiratory capacity, inflammation and neuronal toxicity.”
These latest genetic discoveries confirm a strong link between Lewy body dementia, Parkinson’s disease, and Alzheimer’s disease.
“Interestingly, all five risk loci that we found to be linked to Lewy body dementia have also been shown to either play a role in Alzheimer’s disease or Parkinson’s disease,” Scholz noted.
“What seems to be emerging is that the genetic architecture of Lewy body dementia is quite complex. It is not that a Lewy body dementia patient has all of the genetic features of Alzheimer’s and all of the genetic features of Parkinson’s. Instead, it appears that they overlap only in certain ways, some Alzheimer’s loci here, some Parkinson’s loci there. This is what is so fascinating about this disease, and our study represents an initial step in unraveling this nuance.”
Although Alzheimer’s and Parkinson’s disease are molecularly and clinically very different disorders, she added, “our results support the idea that the problems that cause those diseases may also happen in Lewy body dementia. The challenge we face in treating these patients is determining which specific problems are causing the dementia. We hope studies like this one will help doctors find precise treatments for each patient’s condition.”
The researchers say they hope that shared variants and nodes in common diseases pathways could be targeted for the development of disease-modifying therapies. “Knowing which molecular relationships exist between degenerative diseases will inform clinical trial design and will facilitate the repurposing of drugs for a broad spectrum of diseases,” Scholz suggested.
She concluded: “Our main take-home message from this study is that Lewy body dementia is a genetically complex and polygenic disease that intersects molecularly with Alzheimer’s disease and Parkinson’s disease. We have made the raw data publicly available so that other researchers can access and analyze the data with their own theories in mind. We hope that this will accelerate the pace of discovery in this understudied field.”
Commenting for Medscape Medical News, James Beck, PhD, chief scientific officer at the Parkinson’s Foundation, said this is one of the largest reports of Lewy body dementia genetics.
“This is a really interesting paper which has involved a tremendous amount of work to put together all this genetic information from patients with Lewy body dementia,” noted Beck, who was not involved with the research.
He pointed out that the observations that some of the genes are also linked to Alzheimer’s or Parkinson’s ties in with clinical symptoms seen in the various conditions.
“Lewy body dementia is seen as a condition that fits somewhere in the netherworld between Alzheimer’s and Parkinson’s. We are now recognizing that more specific subtypes of neurodegenerative diseases and genetic discoveries like the ones reported here will help us with this.”
He noted that one of the genes highlighted in this report, SNCA, is an antisense gene for alpha synuclein, the protein at the heart of Lewy body dementia and Parkinson’s disease.
“This antisense gene is likely a control factor for alpha synuclein production and this could be a target for drugs to down-regulate that process,” Beck suggested.
“While it is not a silver bullet, this research is part of the process of developing an information resource that will bring precision medicine to neurodegenerative diseases,” Beck commented. “It gives us directions to identify specific genetic targets. We must think about functionality of various genes and mutations — what they do and can they be modulated?”
He added: “The great hope is that in the future, we may be able to use genetic information in middle age to assess risk of an individual developing various neurodegenerative conditions, and also to develop specific treatments that can slow this process down. We are just at the start of that journey now in that we are beginning to understand how genetics influences neurodegenerative diseases.”
This research was supported in part by the Intramural Research Program of the National Institutes of Health, National Institute on Aging and National Institute of Neurological Disorders and Stroke.
Nature Genetics. Published online February 15, 2021. Abstract
For more Medscape Neurology news, join us on Facebook and Twitter
[ad_2]
Source link












