[ad_1]
Summary: Researchers have identified regions within the DNA of neurons that accumulate single-strand breaks (SSBs). The SSB accumulation appears to be unique to neurons. The findings challenge previous thoughts on the cause of DNA damage and how it could impact neurodegenerative disorders.
Source: NIH
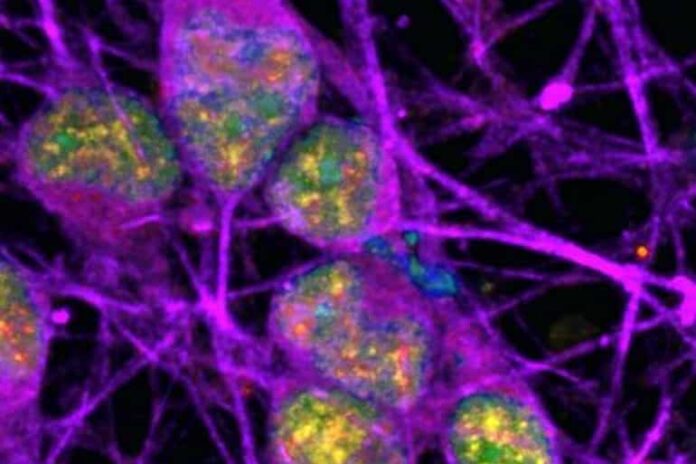
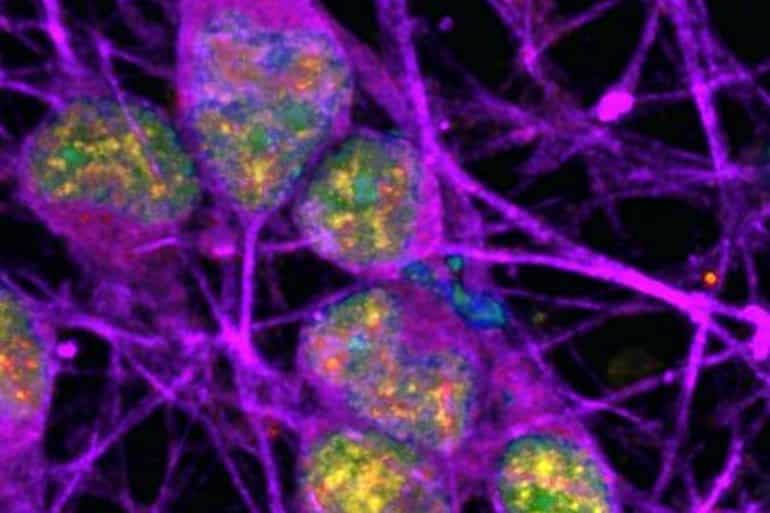
Researchers at the National Institutes of Health (NIH) have discovered specific regions within the DNA of neurons that accumulate a certain type of damage (called single-strand breaks or SSBs). This accumulation of SSBs appears to be unique to neurons, and it challenges what is generally understood about the cause of DNA damage and its potential implications in neurodegenerative diseases.
Because neurons require considerable amounts of oxygen to function properly, they are exposed to high levels of free radicals–toxic compounds that can damage DNA within cells. Normally, this damage occurs randomly. However, in this study, damage within neurons was often found within specific regions of DNA called “enhancers” that control the activity of nearby genes.
Fully mature cells like neurons do not need all of their genes to be active at any one time. One way that cells can control gene activity involves the presence or absence of a chemical tag called a methyl group on a specific building block of DNA. Closer inspection of the neurons revealed that a significant number of SSBs occurred when methyl groups were removed, which typically makes that gene available to be activated.
An explanation proposed by the researchers is that the removal of the methyl group from DNA itself creates an SSB, and neurons have multiple repair mechanisms at the ready to repair that damage as soon as it occurs. This challenges the common wisdom that DNA damage is inherently a process to be prevented.
Instead, at least in neurons, it is part of the normal process of switching genes on and off. Furthermore, it implies that defects in the repair process, not the DNA damage itself, can potentially lead to developmental or neurodegenerative diseases.
This study was made possible through the collaboration between two labs at the NIH: one run by Michael E. Ward, M.D., Ph.D. at the National Institute of Neurological Disorders and Stroke (NINDS) and the other by Andre Nussenzweig, Ph.D. at the National Cancer Institute (NCI). Dr. Nussenzweig developed a method for mapping DNA errors within the genome.
This highly sensitive technique requires a considerable number of cells in order to work effectively, and Dr. Ward’s lab provided the expertise in generating a large population of neurons using induced pluripotent stem cells (iPSCs) derived from one human donor. Keith Caldecott, Ph.D. at the University of Sussex also provided his expertise in single strand break repair pathways.
The two labs are now looking more closely at the repair mechanisms involved in reversing neuronal SSBs and the potential connection to neuronal dysfunction and degeneration.
About this genetics research news
Source: NIH
Contact: Carl P. Wonders – NIH
Image: The image is credited to Ward lab, NINDS
Original Research: Closed access.
“Neuronal enhancers are hotspots for DNA single-strand break repair” by Wei Wu, Sarah E. Hill, William J. Nathan, Jacob Paiano, Elsa Callen, Dongpeng Wang, Kenta Shinoda, Niek van Wietmarschen, Jennifer M. Colón-Mercado, Dali Zong, Raffaella De Pace, Han-Yu Shih, Steve Coon, Maia Parsadanian, Raphael Pavani, Hana Hanzlikova, Solji Park, Seol Kyoung Jung, Peter J. McHugh, Andres Canela, Chongyi Chen, Rafael Casellas, Keith W. Caldecott, Michael E. Ward & André Nussenzweig. Nature
Abstract
Neuronal enhancers are hotspots for DNA single-strand break repair
Defects in DNA repair frequently lead to neurodevelopmental and neurodegenerative diseases, underscoring the particular importance of DNA repair in long-lived post-mitotic neurons. The cellular genome is subjected to a constant barrage of endogenous DNA damage, but surprisingly little is known about the identity of the lesion(s) that accumulate in neurons and whether they accrue throughout the genome or at specific loci.
Here we show that post-mitotic neurons accumulate unexpectedly high levels of DNA single-strand breaks (SSBs) at specific sites within the genome. Genome-wide mapping reveals that SSBs are located within enhancers at or near CpG dinucleotides and sites of DNA demethylation. These SSBs are repaired by PARP1 and XRCC1-dependent mechanisms.
Notably, deficiencies in XRCC1-dependent short-patch repair increase DNA repair synthesis at neuronal enhancers, whereas defects in long-patch repair reduce synthesis. The high levels of SSB repair in neuronal enhancers are therefore likely to be sustained by both short-patch and long-patch processes.
These data provide the first evidence of site- and cell type-specific SSB repair, revealing unexpected levels of localized and continuous DNA breakage in neurons. In addition, they suggest an explanation for the neurodegenerative phenotypes that occur in patients with defective SSB repair.
[ad_2]
Source link