[ad_1]
Summary: Enzyme activity outside of specific cell types contribute to synaptic plasticity associated with addiction behaviors.
Source: Elsevier
Addiction, or substance use disorder (SUD), is a complex neurological condition that includes drug-seeking behavior among other cognitive, emotional and behavioral features. Synaptic plasticity, or changes in the way neurons communicate with one another, drives these addictive behaviors. These lasting brain changes are at the crux of why addiction is so hard to treat.
A new study in Biological Psychiatry, published by Elsevier, now shows that players in the extracellular environment – not just at neuronal interfaces – contribute to addiction plasticity. Neurons in a brain area called the nucleus accumbens are known to undergo addiction-related plasticity. Specifically, changes at synapses of medium spiny neurons (MSN), which sense the neurotransmitter dopamine, have been associated with drug-seeking and extinction behaviors.
Previous research had shown that plasticity at MSNs expressing the D1-type of dopamine receptor is linked to drug-seeking, whereas extinction of drug-seeking involves plasticity at MSNs containing the D2-type dopamine receptor. Now, research led by Peter Kalivas, PhD, and Vivian Chioma, PhD, shows that distinct enzymes working at D1- and D2-type MSNs underlie drug-seeking and extinction behaviors.
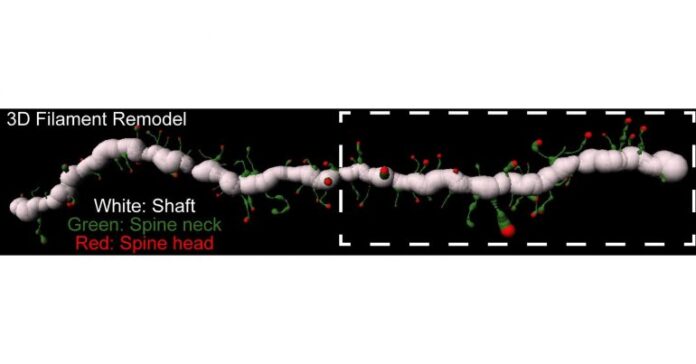
The researchers trained rats to self-administer heroin by pushing a lever for 10 days, followed by a 10-day withdrawal period. They then carefully examined the rats’ brains under a microscope to detect enzymatic activity around the MSN synapses.
The study focused on the activity of extracellular enzymes called metalloproteinases (MMP). MMPs break down proteins that constitute the extracellular matrix around nerve cells. This matrix of proteins supports synaptic connections, but it also constrains the remodeling of synaptic connections in response to experience. Therefore, MMP activity directly impacts cells’ ability to manifest neuroplastic changes.
To assess activity of the MMPs, the researchers used a technique called in vivo zymography, in which the rats’ brains were injected with a fluorescent dye encapsulated in a protective gelatin coat at various points during the drug-training regimen. Once broken open by MMP enzymes, the dye becomes visible, allowing the investigators to observe cells’ morphology as well as the enzymes’ activity around specific cell types and sub-cellular locations.
“We found that drug cues increased MMP activity on one cell type in the nucleus accumbens,” said Dr. Kalivas, referring to D1-type MSNs, “and they decreased activity on another cell type,” the D2-type MSNs. “By showing this cellular specificity of MMP activation and inactivation by cues, we have identified novel molecules that may be potential targets for drug development in treating drug addiction,” he added.
“This paper highlights the exquisite selectivity of addiction-related neuroplasticity,” said John Krystal, MD, Editor of Biological Psychiatry. “In this study, cues associated with heroin delivery activated MMPs near dopamine D1 receptor-containing MSNs in the nucleus accumbens, promoting plasticity in key cells implicated in addiction. Rather than reducing this addiction-related effect on plasticity, extinction training instead increased MMP activity close to neighboring dopamine D2 receptor-containing MSNs – cells implicated in protection against addiction.”
Importantly, the researchers also saw MMP activity associated with cells adjacent to the MSNs called astrocytes, a type of glial cell. The astrocytes, the extracellular matrix, and the two neurons forming a synapse are all part of what is termed the tetrapartite, or four-part, synaptic complex.
“Our work investigating cell-type specific synaptic neuroadaptations in the nucleus accumbens provides evidence of novel advances in our understanding of tetrapartite synaptic activity during heroin seeking,” said Dr. Chioma. “This research demonstrates how integration of components of the tetrapartite synapse regulate specific addiction phenotypes.”
Dr. Krystal added, “This finding suggests that recovery from addiction is not simply a reversal of addiction-related changes in the brain, but rather it also involves the laying down of new anti-addiction changes that protect against substance use.”
About this neuroscience and addiction research news
Source: Elsevier
Contact: Rhiannon Bugno – Elsevier
Image: The image is credited to Elsevier
Original Research: Closed access.
“Heroin Seeking and Extinction from Seeking Activate Matrix Metalloproteinases at Synapses on Distinct Subpopulations of Accumbens Cells” by Vivian C. Chioma et al. Biological Psychiatry
Abstract
Heroin Seeking and Extinction from Seeking Activate Matrix Metalloproteinases at Synapses on Distinct Subpopulations of Accumbens Cells
Table of Contents
Background
Seeking addictive drugs is regulated by synaptic plasticity in the nucleus accumbens core and involves distinct plasticity in D1- and D2-medium spiny neurons (MSNs). However, it is unknown how differential plasticity between the two cell-types is coordinated. Synaptic plasticity and seeking behavior induced by drug-paired cues depends on plasticity not only in the canonical pre- and post-synapse, but also on cue-induced changes in astrocytes and the extracellular matrix adjacent to the synapse. Drug cue-induced signaling in the extracellular matrix is regulated by catalytic activity of matrix metalloproteases-2,9 (MMP-2,9). We hypothesized that the cell-type specific synaptic plasticity is associated with parallel cell-specific activity of MMP-2 and MMP-9.
Methods
Transgenic rats were trained on a heroin self-administration protocol followed by two weeks of drug withdrawal, and a light/tone cue was paired with heroin delivery. Confocal microscopy was used to make morphological measurements in membrane reporter-transduced D1- and D2-MSNs and astrocytes, and MMP-2,9 gelatinase activity adjacent to cell surfaces was quantified using in vivo zymography.
Results
Presenting heroin-paired cues transiently increased MMP-9 activity around D1-MSN dendritic spines and synapse-proximal astroglial processes. Conversely, extinction training induced long-lasting increases in MMP-2 activity adjacent to D2-MSN synapses. Moreover, heroin-paired cues increased tissue inhibitor of metalloproteinases-1,2, which caused transient inhibition of MMP-2 activity around D2-MSNs during cue-induced heroin seeking.
Conclusions
The differential regulation of heroin seeking and and extinguished seeking by different MMP subtypes on distinct cell populations poses MMP-2,9 activity as an important mediator and contributor in heroin-induced cell-specific synaptic plasticity.
[ad_2]
Source link