[ad_1]
Summary: Focusing attention on a neural pathway starting at the periaqueductal grey region of the midbrain, researchers made a novel discovery about how dopamine generates different pain responses in male and female mice. Findings indicate dopamine may reduce pain sensitivity in males, whereas, in females, dopamine helps focus attention elsewhere in the presence of pain.
Source: UNC Health Care
Males and females, generally speaking, experience and respond to pain differently, but scientists have yet to understand all the brain circuits involved in these differences.
Now, new research from the UNC School of Medicine lab of Thomas Kash, PhD, shows how neurons use dopamine to regulate pain differently in male and female mice.
The discovery, published in the journal Neuron, could help the scientific community devise better pain management strategies, particularly for women, who are disproportionally affected by pain throughout their lifespans.
“We focused on this neural pathway because our previous work and that of others show that specific neurons release dopamine to regulate pain responses,” said Kash, the John R. Andrews Distinguished Professor of Pharmacology. “Unfortunately, that research was done only in male mice. So we decided to look at both male and female mice, and what we found was very surprising.”
Dopamine, long known as the brain’s pleasure chemical, can actually regulate a wide variety of behaviors. The dopamine neurons that Kash and his lab looked at had previously been shown to be important for both the rewarding properties and the pain-relieving properties of heroin. Beyond this, several studies have shown that these neurons can regulate attention, suggesting a link between drug abuse, pain, and attention.
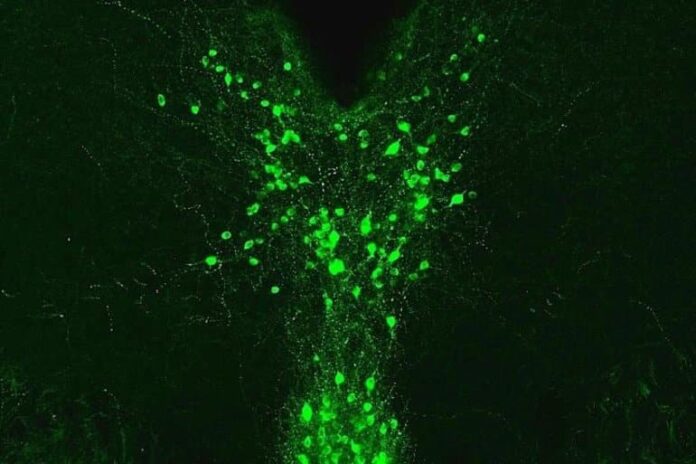
Previously, using male mice, the Kash lab found that dopaminergic neurons played a key role in how opiates dampen pain, likely through the release of dopamine and glutamate. In the new experiments, his lab focused on a neural pathway starting at the midbrain region called the periaqueductal grey, including part of the dorsal raphe.
That brain region is involved in behavioral adaptation – how animals learn to respond to their environment. The neurons that make dopamine in that region operate in conjunction with a brain structure called the bed nucleus of the stria terminalis, or BNST, forming a neural pathway.
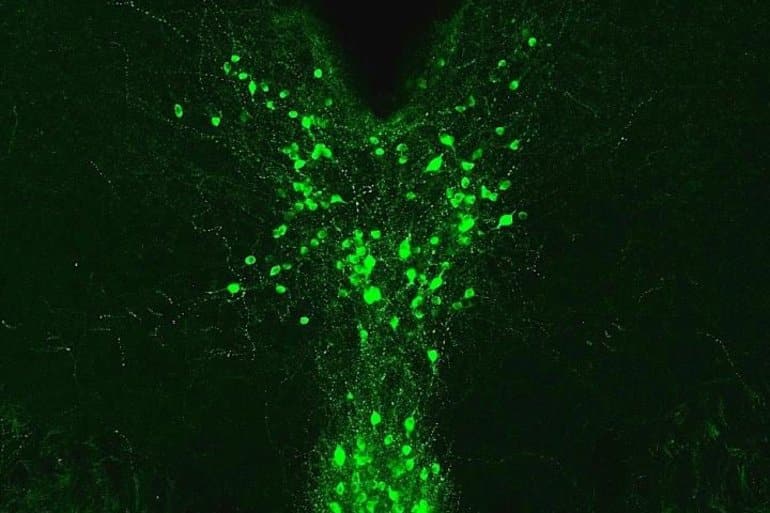
“We found that activating this pathway reduced pain sensitivity in male mice, but made female mice move more, especially in the presence of something capturing their attention,” said first author Waylin Yu, PhD, a former graduate student in the Kash lab and current postdoctoral researcher at UC San Francisco. “We think this is because of the different ways males and females respond to pain.”
In particular, these experiments seem to indicate that dopamine helps males simply not feel as much pain, while in females, dopamine helps the mice focus attention elsewhere while in the presence of pain.
More research is needed, but the Kash lab research shows that activating specific neural projections to the BNST reduces acute and persistent inflammatory pain, providing further evidence that dopamine signaling can enhance the blocking of pain stimuli, thus counteracting severe pain.
“We hope to investigate how this pathway can regulate more emotional behaviors associated with chronic pain, and then also look at the dynamics of the system, such as how this pathway works in real time during behavior measurements,” Kash said. “These neurons are also implicated in the actions of opioids such as morphine, so we plan to investigate that domain, as well.”
Funding: The National Institutes of Health funded this research.
Other authors on the Neuron paper are Dipanwita Pati, PhD, postdoctoral fellow; Melanie Pina, PhD, postdoctoral fellow; Karl Schmidt, PhD, a postdoctoral fellow in the UNC Department of Psychiatry; Kristin Boyt, senior research analyst in the Kash lab; Avery Hunker, PhD, and Larry Zweifel, PhD, both at the University of Washington; and Zoe McElligott, PhD, assistant professor of psychiatry at the UNC School of Medicine. Thomas Kash is a member of the UNC Bowles Center for Alcohol Studies.
Table of Contents
About this pain research news
Source: UNC Health Care
Contact: Mark Derewicz – UNC Health Care
Image: The image is credited to Kash Lab, UNC School of Medicine
Original Research: Closed access.
“Periaqueductal gray/dorsal raphe dopamine neurons contribute to sex differences in pain-related behaviors” by Thomas Kash et al. Neuron
Abstract
Periaqueductal gray/dorsal raphe dopamine neurons contribute to sex differences in pain-related behaviors
Highlights
- •vlPAG/DRDA to BNST activation promotes anti-nociception in male mice
- •vlPAG/DRDA to BNST activation promotes locomotion in female mice
- •Membrane-gated currents driven by optically evoked DA release differ by sex
- •Dopamine is required for vlPAG/DR-driven anti-nociception
Summary
Sex differences in pain severity, response, and pathological susceptibility are widely reported, but the neural mechanisms that contribute to these outcomes remain poorly understood.
Here we show that dopamine (DA) neurons in the ventrolateral periaqueductal gray/dorsal raphe (vlPAG/DR) differentially regulate pain-related behaviors in male and female mice through projections to the bed nucleus of the stria terminalis (BNST).
We find that activation of vlPAG/DRDA+ neurons or vlPAG/DRDA+ terminals in the BNST reduces nociceptive sensitivity during naive and inflammatory pain states in male mice, whereas activation of this pathway in female mice leads to increased locomotion in the presence of salient stimuli.
We additionally use slice physiology and genetic editing approaches to demonstrate that vlPAG/DRDA+ projections to the BNST drive sex-specific responses to pain through DA signaling, providing evidence of a novel ascending circuit for pain relief in males and contextual locomotor response in females.
[ad_2]
Source link